“On the feature list of health and fitness wearables, heart rate (HR) and blood oxygen saturation (SpO2) are rapidly moving from “desirable” to “hopeful”. However, the shift has led to a decline in the quality of readings, as some sensor manufacturers have relaxed the quality of readings on their products in a rush to meet market demand for these features, raising questions about the accuracy of their products.
“
On the feature list of health and fitness wearables, heart rate (HR) and blood oxygen saturation (SpO2) are rapidly moving from “desirable” to “hopeful”. However, the shift has led to a decline in the quality of readings, as some sensor manufacturers have relaxed the quality of readings on their products in a rush to meet market demand for these features, raising questions about the accuracy of their products.
While reading accuracy may be less critical in everyday wearables, in clinical-grade wearables the quality and integrity of the measurements must be unquestionable. Designers face a key challenge: how to make high-quality HR and SpO2 measurements without draining the device battery too much? In this design solution, we first show why traditional optical readout methods waste power, and then introduce a sensor IC with a novel architecture that can perform clinical-grade measurements while significantly reducing power consumption.
Photoplethysmography (PPG)
We measured HR and SpO2 using an optoelectronic technique called photoplethysmography, or PPG (Figure 1). The PPG signal is obtained by detecting changes in the intensity of light reflected from blood vessels below the surface by illuminating the skin with a light-emitting diode (LED) and using a photodiode (PD) to generate a current proportional to the amount of light received (Figure 2).

Figure 1. Measurement of HR and SpO2 using a wrist-worn device.

Figure 2. PPG measurement using LED and PD.
The current signal is conditioned by the PPG analog front end (AFE) and then converted by the ADC for processing by an optical algorithm running on the system microcontroller. In principle, a single pair of LED-PDs is sufficient for PPG measurements, a configuration that is common in clinical devices (Fig. 3).

Figure 3. Measurement of SpO2 and HR in a clinical setting.
However, these devices operate in a completely different environment than in everyday life. First, the patient remains still and measurements are taken by sensors clipped to the patient’s fingertips. The lighting conditions are relatively stable, which simplifies the light detection of PDs, and these devices are generally powered by the mains supply, so there is no need to worry about power consumption.
In contrast, wearables are typically worn on the wrist, which means that the level of contact with the skin generally depends on personal preference (how tight the wristband is) and the movements of the wearer. Lighting conditions can vary greatly from day to day with location and time of day, and these devices are battery powered, so it is imperative to keep the sensor’s current consumption as low as possible. Also, different wearers have different skin tones, which makes the problem even more challenging. According to the description, the perfusion index of dark skin is lower than that of light skin, which means that to make the measurement, a greater illumination intensity is required (requiring the sensor to consume more power). Next, we look at the advantages of different AFE structures for making PPG measurements.
PPG AFE with single ADC channel
Increasing the LED current or using two LEDs is a very intuitive way to increase the intensity of the skin irradiation (Figure 4), which increases the skin’s irradiated area. However, this approach is very power-intensive, as the LED current accounts for at least 50% of the total power consumption of the PPG system, which may average 1 mW depending on the wearer’s skin perfusion index. Overall, this approach is inefficient and bad for battery life.

Figure 4. Using two LEDs to increase skin illumination intensity.
PPG AFE with two ADC channels
A better approach we can use to increase the skin exposure is to use an LED containing two PDs, which can be used to detect more reflected light (Figure 5).

Figure 5. Using an LED containing two PDs to improve light detection.
The advantage is that the standard 20 mA LED current can be reduced to 10 mA compared to using a single PD, resulting in the same level of total PD current. Under challenging operating conditions (low skin perfusion and/or when the wearer is moving), the system’s algorithm determines that higher LED current is required, and the system sensitivity increases proportionally. For example, with the same LED current as before, twice the PD current is provided, which results in higher overall sensitivity, albeit at an increased power cost.
PPG AFE with Four ADC Channels
Using four PDs (requiring a four-channel ADC) to detect reflected light saves more power (Figure 6) because the LEDs can operate at lower power (Table 1).
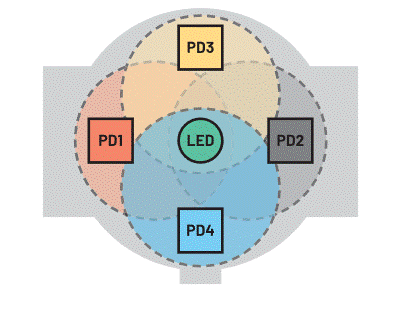
Figure 6. PPG measurement using one LED and four PDs.

Table 1. Typical Power Consumption Comparison for 1-, 2-, and 4-Channel ADC Architectures
Table 1 summarizes the relative power dissipation of the various structures considered previously, assuming a typical supply voltage of 1.6 V.
This configuration provides higher-quality readings due to the asymmetric distribution of blood vessels and bones in the wrist, but the four PDs can help eliminate the effects of motion, as well as the tightness of the device. The four PD receivers also increase the probability of detecting reflected light from the illuminated vessels. The graph in Figure 7 shows the HR measured using 4 photodiodes (configured as two independent pairs: LEDC1 and LEDC2) compared to a reference measurement (polar). Wearable devices need to ensure good skin-to-skin contact during measurements. Initially, the wearer rested and started exercising after 5 minutes (300 seconds), causing their HR to start to rise. Obviously, the signals on LEDC1 and LEDC2 deviate from the reference measurement by different degrees, so it is beneficial to use two pairs of PDs to capture the signal and account for all these deviations in combination.

Figure 7. HR readings obtained when using two independent pairs of PDs.
Practical Quad ADC Solution
The MAX86177 (Figure 8) is an ultra-low-power quad-channel optical data-acquisition system with transmit and receive channels ideal for clinical-grade (and general-purpose) portable and wearable devices. The transmitter integrates two high-current 8-bit programmable LED drivers, supporting up to 6 LEDs. The receiver integrates four low-noise charge integration front ends, each containing an independent 20-bit ADC that can multiplex input signals from eight PDs (configured as four independent pairs). It achieves a dynamic range of 118 dB and provides up to 90 dB of ambient light cancellation (ALC) at 120 Hz. The main supply voltage is 1.8 V, and the LED driver supply voltage is 3.1 V to 5.5 V. The device provides full autonomous support for I2C and SPI compatible interfaces. The MAX86177 is available in a 2.83 mm × 1.89 mm, 28-pin (7 × 4) wafer-level package (WLP) and operates over the C40°C to +85°C temperature range. A laboratory test sample of this AFE showed a total root mean square error of 3.12% for hypoxia measurements, which was within the 3.5% limit set by the FDA for clinical-grade monitors.
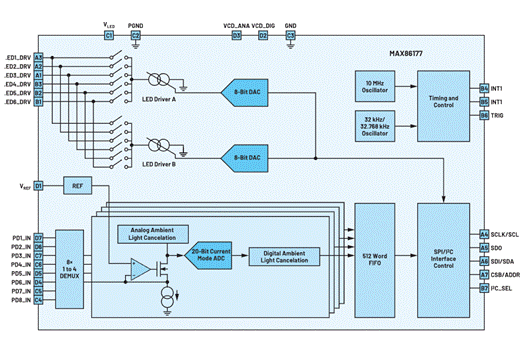
Figure 8. Block diagram of the MAX86177 quad optical AFE.
in conclusion
A major challenge for bed-level wearable device designers is how to perform optical PPG measurements to obtain HR and SpO2 values without significantly draining the device’s battery life. In this design solution, it can be seen that the four-channel ADC structure can save up to 60% of power compared to the basic structure using a single LED and PD. The MAX86177’s quad-channel architecture in a small package is ideal for finger, wrist, and ear-worn wearables for clinical-grade HR and SpO2 measurements. It can also be used to measure body water content, muscle and tissue oxygen saturation (SmO2 and StO2), and maximum oxygen consumption (VO2 max).